Article dedicated to Dr. Adolfo Garcia Sastre, the “Doctor Bacteria” of the 21st century and his Scientific School, all of them committed Cajalians. Adolfo is an Honorary Member of Alumni, a Distinguished Guest of the city of Salamanca, among many other well-deserved honors.
“The indescribable pleasure — which dims all other joys of life — is an abundant compensation for the researcher who endures the painful and persevering analytical work that precedes the emergence of the new truth, like the pain of childbirth. It is true to say that nothing for the scientific scholar is comparable to the things he has discovered” (“El placer indescriptible — que empalidece el resto de las alegrias de la vida — es una abundante compensacion para el investigador que soporta el doloroso y perseverante trabajo analitico que precede a la aparicion de la nueva verdad, como el dolor del parto. Es cierto decir que nada para el erudito cientifico es comparable a las cosas que ha descubierto”)
Santiago Ramon y Cajal
In 1903, Santiago Ramon y Cajal published in the Trabajos del Laboratorio de Investigaciones Biologicas an article that, on the surface, appeared purely technical: “A Simple Method of Selective Staining of the Protoplasmic Reticulum” (“Un sencillo metodo de coloracion selectiva del reticulo protoplasmatico”). In it, he detailed with his characteristic meticulousness a new silver nitrate impregnation method that promised consistent results applicable to the entire nervous system. However, among the descriptions of solutions and fixation times, his tireless gaze — trained to discern order in chaos — paused on a minuscule detail that had gone unnoticed by everyone. When describing the nucleus of neurons, he noted: “…all neurons, without exception, exhibit another special spherical corpuscle, exceedingly small, generally unique… Such granules, which for want of a better name we shall call accessory intranuclear bodies” (“…todas las neuronas, sin excepcion, exhiben otro corpusculo especial esferico, pequeñisimo, generalmente unico… Semejantes granitos, que por llamarlos de algun modo los denominaremos cuerpos intranucleares accesorios”).
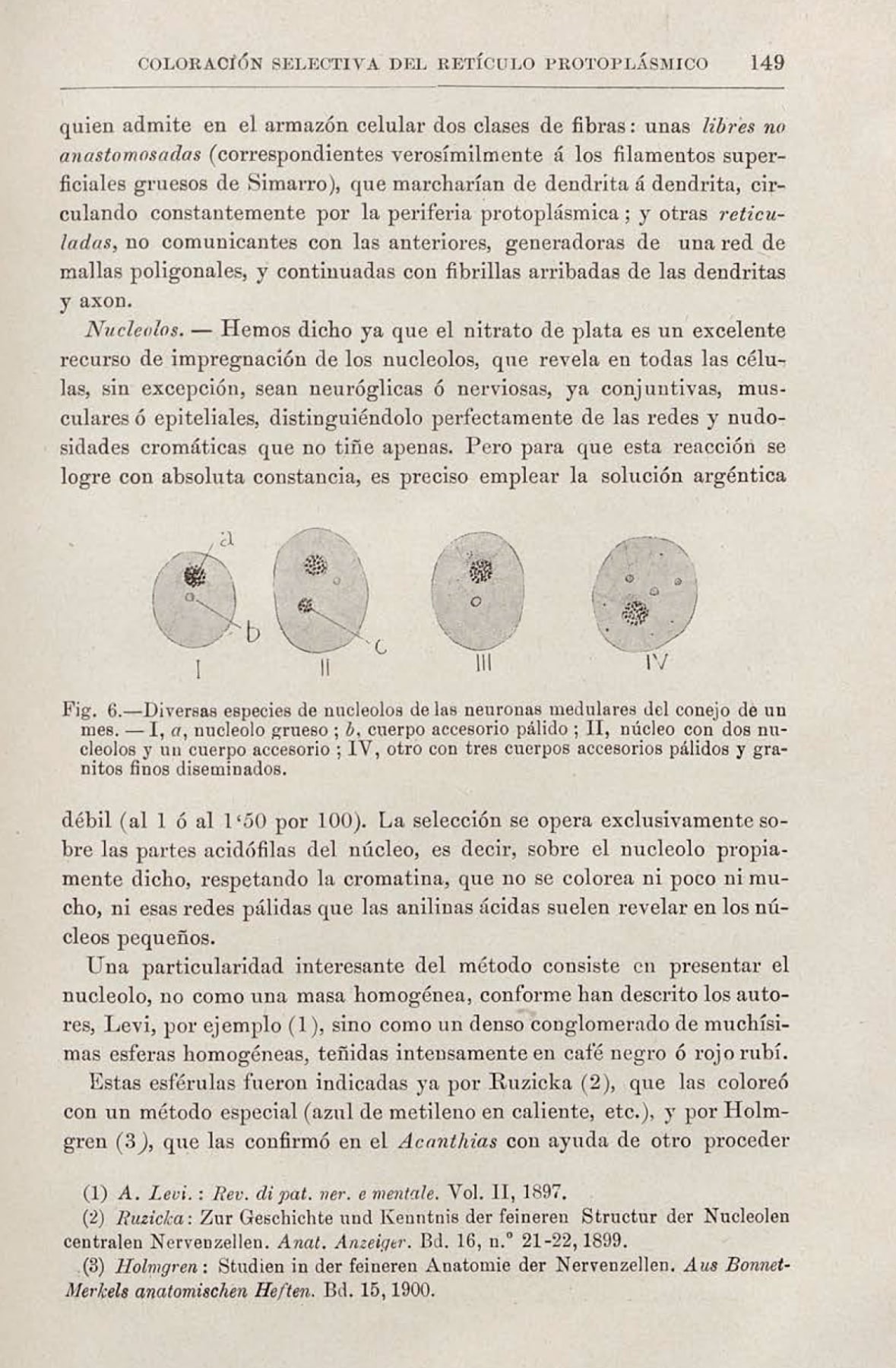


He observed that these small spheres stained a “yellow or orange” tone, distinct from the black of the nucleolus, and concluded that their chemical composition “is not identical.” This discovery was not an accident, but the inevitable consequence of his singular scientific method: a sublime fusion of rigorous empirical observation and a profound, almost mystical, artistic sensibility.
The Eye of the Artist, the Mind of the Scientist
Cajal fervently maintained that every neuroanatomist should be an artist, for the act of drawing was not a mere record, but a tool of inquiry, a form of visual thinking. Unlike cold and impartial photography, drawing allowed the scientist-artist to synthesize multiple observations, interpret three-dimensional architecture, and — most importantly — discern order in the chaos of the “tangled and mysterious forest” (“enmaranado y misterioso bosque”) of the nervous system. It was precisely his new staining method, detailed in his 1903 publication (article appended at the end of this article), that granted him the clarity of vision necessary to identify structures that had gone unnoticed by others. This “discipline of the eye,” cultivated through drawing, was not a skill for recording, but for understanding. Cajal’s ability to abstract and construct a coherent mental model from fragmentary data was the same ability that allowed him to see neurons as individual entities — overturning Golgi’s reticular theory — and that enabled him to identify these subtle corpuscles that others, with a less poetic gaze, might have dismissed as mere artifacts. His art was not a byproduct of his science; it was the tool that made it possible.
This perspective holds profound resonance in contemporary science. In an era dominated by “big data” and imaging technologies of unprecedented resolution, the human capacity to interpret, conceptualize, and find patterns — the “eye of the artist” — remains irreplaceable. Cajal’s legacy teaches us that true understanding does not emerge from the mere accumulation of data, but from its meaningful interpretation. More than a century later, that observation — born of the confluence between art and science — has blossomed into a field of critical and surprising importance. The “accessory body,” today universally known as the Cajal Body, reveals itself as a strategic epicenter, a central battlefield in the ceaseless arms race between the host cell and invading viruses. This article explores how Cajal’s hidden legacy has transitioned from a cytological curiosity to the vanguard of virology and nuclear immunity, tracing a direct line from the philosophy of the Spanish sage to the laboratories that today combat the pandemics of the future.
A Condensate of Life at the Heart of the Nucleus
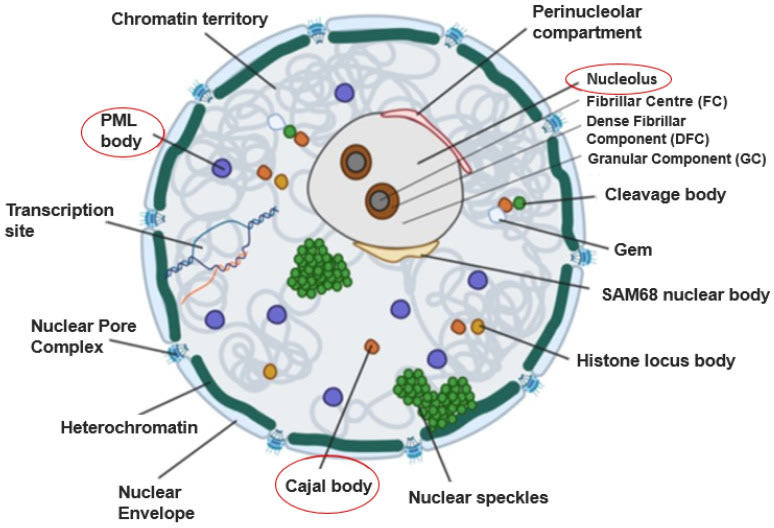
The structure that Cajal described as an appendage of the nucleolus has been radically redefined by molecular biology. Today we know that the Cajal Body (CB) is not a static organelle, but an architecture of life: a dynamic, membraneless structure classified as a “biomolecular condensate.” These condensates are comparable to ephemeral droplets that self-organize within the nucleoplasm, formed by a physical process known as liquid-liquid phase separation. Their primary function is to concentrate specific proteins and RNA in an infinitesimal space, drastically accelerating the efficiency of biochemical reactions without the need for a physical barrier such as a membrane.
Coilin: The Architect Protein
The identity and very existence of the Cajal Body depend on a key protein: p80-coilin. Coilin acts as an essential scaffolding protein; its ability to interact with itself and to bind a multitude of other molecules is what initiates the formation of the condensate. Although it is present diffusely in the nucleoplasm, its enrichment in these bodies is what defines them. The regulation of this process is exquisitely complex, managed through post-translational modifications of coilin itself — such as phosphorylation, methylation, and SUMOylation — which function as molecular switches to control its interactions, its stability, and ultimately, the assembly and disassembly of the Cajal Body in synchrony with the cell cycle. For example, CBs disassemble during mitosis and reassemble at the onset of interphase, a process regulated by the phosphorylation of coilin, which protects it from degradation.
A Crucible for Genetic Information
The primary function of the Cajal Body places it at the very heart of gene expression. It is a nerve center for the biogenesis, maturation, and modification of small nuclear ribonucleoproteins (snRNPs), the molecular components of the splicing machinery. Splicing is the crucial editing process by which non-coding sequences (introns) are removed from messenger RNA (mRNA) transcripts before they can be translated into proteins. The CB functions as an “assembly line” and a “quality control center” where snRNPs are assembled, modified (through processes such as 2’-O-methylation and pseudouridylation guided by small Cajal body-specific RNAs, or scaRNAs), and prepared for their vital task.
The physics of phase separation is not a mere structural detail; it is the direct cause of the CB’s function. By creating a microenvironment that concentrates reagents, the CB overcomes the limitations of diffusion in the nucleoplasm and accelerates biochemical reactions, acting as a high-efficiency RNP factory. This efficiency is precisely what makes it indispensable for the cell and, therefore, a priority target for viruses. The CB’s vulnerability to viral manipulation is not a coincidence, but a direct consequence of its fundamental operating mechanism. Viruses have evolved to exploit the cell’s point of greatest efficiency, turning the CB into a dynamic computational node that, if controlled, allows domination of the flow of cellular genetic information.
The Nuclear Archipelago: What Did Cajal Really See?
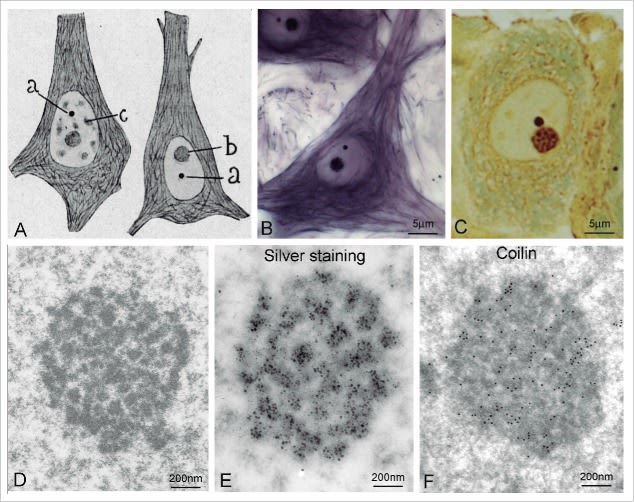
Cajal’s visual acuity — capable of isolating the “accessory body” from the rest of the structures — leads us to a fascinating question: was he also the first to glimpse other nuclear bodies, such as PML Bodies, decades before their formal description? A detailed analysis of his drawings, such as the one shown below, offers us a nuanced answer. Recuerdos de mi vida > Table of Contents > Part Two, XXIV.
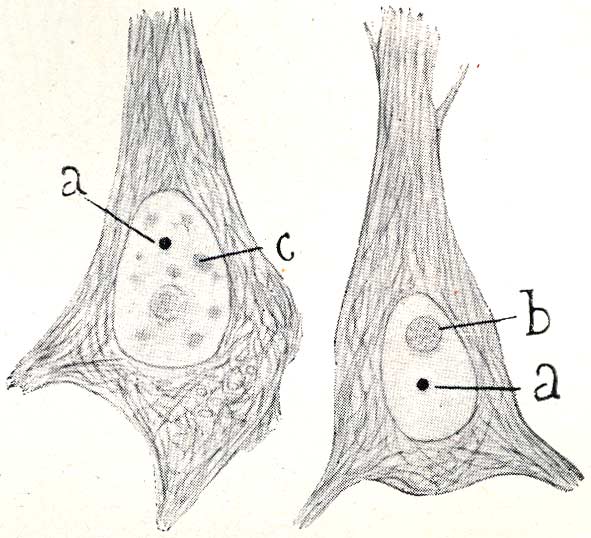
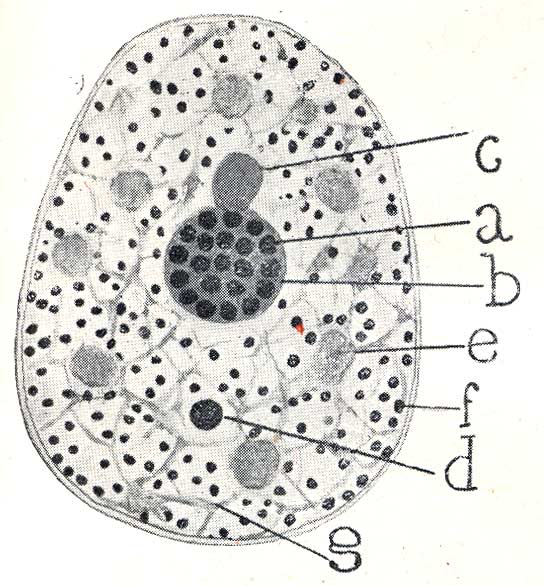

This drawing is irrefutable proof that Cajal saw a universe of nuclear bodies, not just one. With astonishing precision, he not only labels the nucleolus (‘a’) and his “accessory body” (‘b’), but catalogs a complete ecosystem of structures: the “chromatic cap” (‘c’), the “hyaline clump” (‘e’), the “basophilic granules” (‘f’), and the “fibrillar framework” (‘g’). This demonstrates that his gaze penetrated the complexity of the nucleus to a level that no one had achieved.
It is highly probable that some of the various “granules” and “clumps” he drew were, in fact, what we now know as PML Bodies. However, he did not group them under a single denomination nor describe them as a singular and recurrent structure, unlike what he did explicitly with the “accessory body.” The distinction we make today between a Cajal Body, a PML Body, or a nuclear speckle is fundamentally molecular, defined by their scaffolding proteins (Coilin for the CB, PML protein for the PML body, etc.). Cajal’s staining method, based on silver nitrate, was a chemical technique that revealed morphology, not protein composition.
In summary, this drawing does not allow us to claim that Cajal “discovered” PML Bodies, but it demonstrates in a spectacular manner that he was the first to chart the unknown archipelago of the cell nucleus. He saw the different “islands,” even though he did not have the tools to give a geological name to each one. It is, once again, proof of his visionary genius.
The Human Connection: SMN and Spinal Muscular Atrophy (SMA)
The critical importance of the Cajal Body for human health is dramatically illustrated by its connection to Spinal Muscular Atrophy (SMA), a devastating neurodegenerative disease. SMA is caused by the deficiency of the Survival of Motor Neuron (SMN) protein, a fundamental component that localizes prominently in CBs. The SMN protein is part of an essential complex for the assembly of snRNPs in the cytoplasm. Once assembled, the snRNPs, likely associated with SMN, are imported into the nucleus and directed to CBs for their final maturation.
The SMN protein interacts directly with coilin, and this interaction is crucial for the proper assembly and integrity of the CB. In the absence of coilin, neither SMN nor snRNPs localize correctly in the residual CBs. In SMA patients, the lack of functional SMN leads to the disruption and loss of CBs in motor neurons, resulting in defective assembly of the splicing machinery, progressive paralysis, and in the most severe cases, death. The loss of Cajal Bodies is, therefore, a distinctive cellular hallmark of SMA, establishing an unequivocal link between the integrity of this organelle and neuronal survival.
Echoes of a Forgotten Past: Cajal, the Microbiologist
Although history remembers Cajal as the father of neuroscience, his career spanned much broader fields, including a decisive foray into microbiology and public health. This often-forgotten facet establishes a powerful parallel between his historical fight against pathogens and the cellular battle being waged today inside the Cajal Body. In 1885, a devastating cholera epidemic ravaged Spain, and Cajal, then a professor in Valencia, immersed himself in the study of the disease. His work was not limited to observation; he developed an innovative “chemical vaccine” using heat-inactivated cholera bacillus cultures. This approach, which introduced the concept of using dead microbial components to generate immunity, was pioneering and anticipated similar, more recognized work by American researchers.

His commitment to the fight against infectious diseases transcended the laboratory. In 1900, Cajal was appointed first director of the Instituto de Sueroterapia, Vacunacion y Bacteriologia de Alfonso XIII, which would later become the Instituto Nacional de Higiene. He accepted the position out of “patriotic responsibility” (“responsabilidad patriotica”), sacrificing his beloved personal research to provide Spain with health sovereignty in the production of sera and vaccines. His management was a titanic battle against political indifference and bureaucracy. The institute’s first headquarters was a former unsanitary dairy barn, and Cajal found himself consumed by what he himself called the “tyranny of stamped paper” (“tirania del papel timbrado”), going so far as to advance money from his own pocket to avoid halting the vital production of vaccines.
This stage of his life establishes a profound thematic resonance with the central theme of this article. Cajal’s struggle to establish a national health defense infrastructure is a reflection, at the social scale, of the cell’s struggle to mount an antiviral defense at the subnuclear level. The Institute of Hygiene was the “cornerstone of Spanish public health” (“piedra angular de la salud publica espanola”); the Cajal Body is a cornerstone of RNA processing and, as we shall see, of nuclear defense. The symmetry is astonishing: the historical Cajal waged a macroscopic war against a bacterium; current science unveils a microscopic war against viruses that takes place in the very organelle he discovered.
The Nuclear Battlefield: The Hijacking of Cajal’s Legacy
The fundamental importance of the Cajal Body in RNA processing has not gone unnoticed by viruses. Throughout evolution, a broad range of viruses, both DNA and RNA, have developed convergent strategies to hijack this nuclear structure. Their goal is to subvert the nuclear architecture and the host’s RNA processing machinery to put it at the service of their own replication. Viruses do not merely attack the CB; they strategically remodel it, treating it as a programmable resource.
The fact that viruses from families as diverse as Adenoviridae, Orthomyxoviridae, and Flaviviridae converge on the CB as a target is irrefutable proof of the centrality of this organelle. It is not a casual target, but a fundamental “bottleneck” in cell control, a nexus where viruses can prioritize their own RNAs and destabilize those of the host. For this reason, the state of the Cajal Body can be considered a biomarker of viral infection, a nuclear “canary in the coal mine.”
Viral Hijacking Tactics
These viral manipulations cause drastic changes in the morphology, number, and distribution of Cajal Bodies, visible under the microscope. The arsenal of hijacking tactics is diverse and sophisticated:
-
Fragmentation: Viruses such as influenza A or Zika cause CBs to break into multiple smaller and more numerous bodies, dispersing their components.
-
Reorganization: Adenovirus reorganizes CBs into unique structures called “microfoci” or “rosettes,” which position themselves strategically alongside the viral DNA replication centers.
-
Creation of New Bodies: Some viruses, such as the Minute Virus of Mice (MVM), go a step further and co-opt CB components to build entirely new viral replication factories.
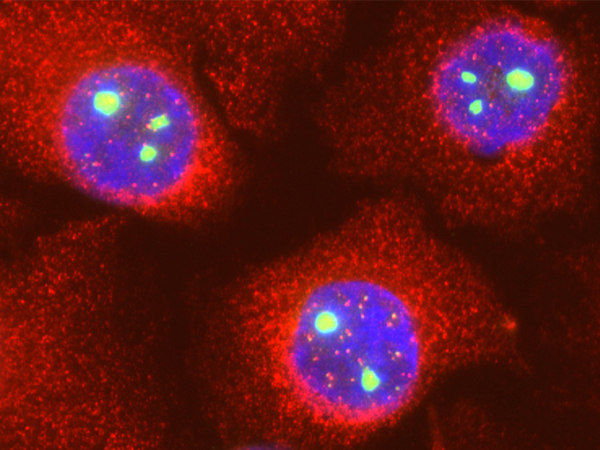
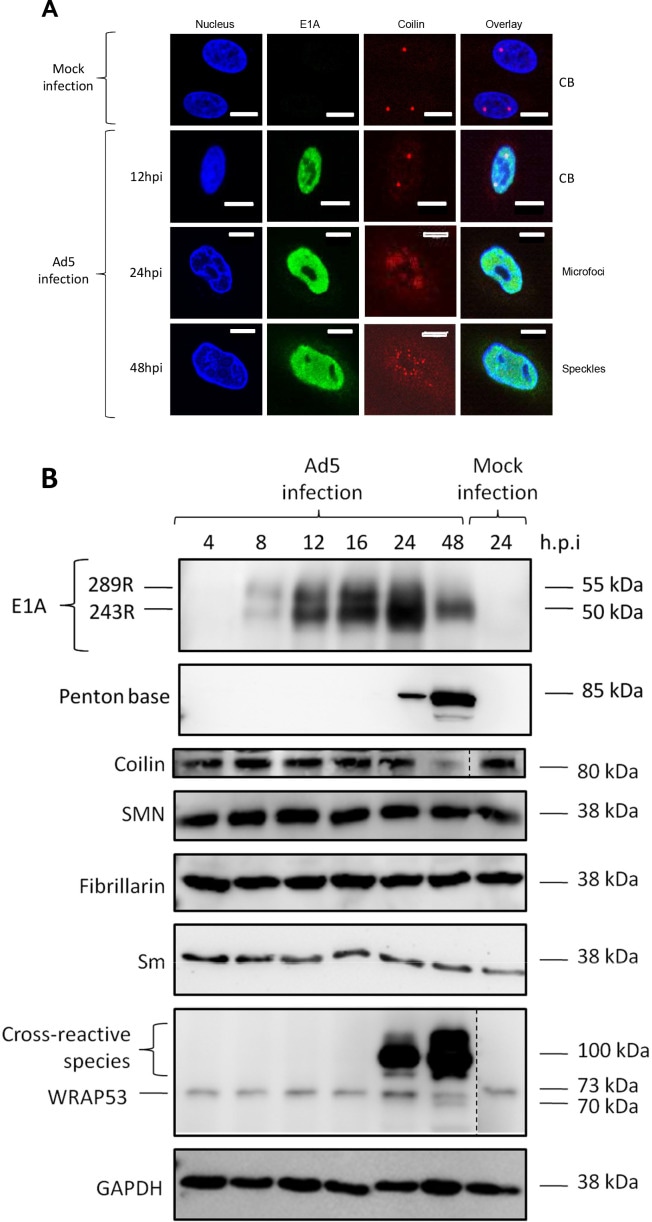
The case of Adenovirus provides a paradigmatic example of this molecular coup d’etat. The viral protein L4-22K binds directly to and forms a stable complex with the host’s coilin. This hijacking serves a very specific purpose: to use coilin as a “safe-conduct pass” to facilitate the export of viral messenger RNAs from the nucleus to the cytoplasm, an indispensable step for the synthesis of new viral proteins. Coilin depletion drastically reduces the virus’s yield by preventing the export of its mRNAs. In essence, the virus turns the chief architect of the CB into an accomplice for executing its own genetic blueprints, as illustrated in Figure 6, which shows the dramatic reorganization of coilin into microfoci following Ad5 infection.
The following table summarizes the diversity of strategies that different viruses employ to manipulate the Cajal Body, illustrating the central nature of this organelle in the virus-host conflict.
| Viral Family/Example | Morphological Change in CBs / Key Molecular Mechanism / Consequence for Viral Infection / Adenovirus (Ad5) |
|---|---|
| Influenza A Virus | Increase in number, decrease in size. Colocalization with viral nucleoprotein (NP). Interaction with viral NP. Possibly related to the trafficking of viral components or subversion of the splicing machinery. |
| Zika Virus (ZIKV) | Increase in number and reduction in volume. The viral protein NS5 interacts with coilin, causing CB disruption. Subversion of nuclear architecture to favor viral replication. |
| Minute Virus of Mice (MVM) | Reconstitution of CB components into new viral factories (APAR bodies). Co-optation of CB components, including the SMN protein. Creation of dedicated and efficient viral replication centers. |
| Peanut Rosette Virus (GRV) | Reorganization into “CB-like” structures that fuse with the nucleolus. The viral protein ORF3 mediates the reorganization and fusion with the nucleolus. Hijacking of cellular components for viral movement and RNA stabilization. |
The Cellular Counteroffensive: The Antiviral State and the Mx Guardians
Faced with this invasion, the cell does not remain passive. It has developed a sophisticated, multi-layered defense system, and at the heart of the innate response lies a family of guardian proteins whose history is intertwined with the fight against the very virus that manipulates the Cajal Body: influenza.
The Alarm Signal: The Interferon System
The cell’s first line of defense is the detection of the intruder. The presence of pathogen-associated molecular patterns (PAMPs), such as double-stranded viral RNA, activates cellular sensors that trigger a signaling cascade. The result is the production and secretion of specialized cytokines called type I (alpha/beta) and type III (lambda) interferons (IFN). These molecules act as a molecular alarm siren. They bind to receptors on the surface of the infected cell (autocrine action) and on neighboring uninfected cells (paracrine action), inducing the expression of hundreds of interferon-stimulated genes (ISGs). This process establishes a potent “antiviral state” that prepares cells to combat infection, creating an inhospitable environment for viral replication.
The Guardians of the Genome: Mx Proteins (Mx1/MxA)
Among the most potent and best-studied effectors of this ISG arsenal is the family of myxovirus resistance (Mx) proteins. Discovered more than 50 years ago thanks to the observation that a strain of mice (A2G) was naturally resistant to influenza virus infection, Mx proteins are large GTPases belonging to the dynamin superfamily. Their expression is strictly dependent on type I and III interferons, and their antiviral function is autonomous: they can protect cells from infection even in the absence of other ISGs. Two properties are absolutely essential for their function: their ability to hydrolyze GTP (GTPase activity) and their ability to self-assemble by forming oligomers, such as rings and helices.
A Localization Strategy: Nuclear vs. Cytoplasmic Defense
The efficacy of Mx proteins lies not solely in their biochemistry, but in an interdependent triad of localization, oligomerization, and GTPase activity. Dysfunction in any of these three pillars abolishes their defensive capacity. Subcellular localization is a key determinant of their spectrum of action. The mouse Mx1 protein, the prototype of influenza resistance, accumulates predominantly in the nucleus. By contrast, its human homolog, the MxA protein (encoded by the MX1 gene), is almost exclusively cytoplasmic. This evolutionary divergence has led to functional specialization: Mx1 is a nuclear guardian, ideally positioned to intercept viruses that, like influenza, must replicate their genome in the nucleus. MxA, on the other hand, acts as a cytoplasmic sentinel, intercepting viral components before they reach their nuclear destination, which confers upon it a broader antiviral spectrum against viruses that replicate in different cellular compartments.
| Key Characteristic | Mx1 (Mouse) / MxA (Human) / Prototype Species |
|---|---|
| Gene | *Mx1 |
| MX1 | *Cellular Localization |
| Predominantly Nuclear / Predominantly Cytoplasmic | Antiviral Spectrum |
| Specialist, potent against viruses with nuclear replication (e.g., Influenza, Thogoto). / Generalist, broad spectrum against RNA viruses and some DNA viruses. | Mechanism vs. Influenza |
| Blocks primary transcription of the viral genome within the nucleus. / Intercepts viral ribonucleoprotein (vRNP) complexes in the cytoplasm, preventing their nuclear import or subsequent function. | Role as Barrier** Model key for studying innate resistance to influenza in mammals. / Acts as an interspecies barrier against the transmission of zoonotic viruses (e.g., avian influenza). |
The Mechanism of Action Against Influenza: Disarming the Viral Replication Complex
The target of Mx proteins in influenza infection is the very heart of the virus: the viral ribonucleoprotein complex (vRNP). This complex is formed by the viral RNA genome, which is coated by multiple copies of the nucleoprotein (NP) and associated with the viral polymerase (composed of the subunits PB1, PB2, and PA). Mx proteins specifically recognize NP within this complex. Following recognition, Mx molecules utilize their self-assembly capacity to oligomerize and form ring-like structures that surround and trap the vRNP. GTP hydrolysis likely drives conformational changes that “tighten the trap,” destabilizing the viral complex and preventing its function. Specifically, Mx1 has been shown to directly interfere with the crucial interaction between NP and the polymerase subunit PB2, an indispensable step for the transcription and replication of the viral genome.
The Evolutionary Arms Race
The human MxA protein is not merely a restriction factor; it is an evolutionary guardian that defines, in part, the species barrier for influenza viruses. It is remarkably effective at restricting avian influenza viruses, such as H5N1, representing a significant obstacle to their transmission to humans. However, viruses that have managed to establish themselves in the human population and cause pandemics, such as the 1918 strain and H1N1 of 2009, did so by acquiring adaptive mutations in their NP protein. These mutations alter the surface of NP at the MxA recognition sites, allowing the virus to evade this first line of innate defense. Therefore, surveillance of NP sequences of influenza viruses circulating in animal reservoirs — with a focus on the regions that interact with MxA — becomes a public health tool of critical importance for predicting the pandemic potential of new strains.
An Ecosystem of Defense: The Architecture of Nuclear Immunity
The antiviral response does not depend on isolated effectors such as Mx proteins, but on a coordinated network and an architectural reorganization of the nucleus. Interferon signaling not only induces the expression of hundreds of genes, but remodels the nuclear landscape, strengthening and connecting functional compartments to create an integrated and resilient defense network.
PML Bodies as Antiviral Command Centers
Another key actor in this defense are the Promyelocytic Leukemia Bodies (PML bodies), also known as Nuclear Domain 10 (ND10). Like CBs, they are membraneless biomolecular condensates whose formation and size increase dramatically in response to interferon and other cellular stresses. PML bodies act as “headquarters” or antiviral command centers. They are sites of intense post-translational modification, especially SUMOylation, a process in which the small protein SUMO (Small Ubiquitin-like Modifier) is added to other proteins. This modification can alter the localization, stability, or activity of its targets, and PML bodies serve as “SUMOylation factories” that activate and regulate numerous viral restriction factors and DNA damage response proteins.
Collaboration Between Nuclear Bodies
These command centers do not operate in isolation. There is evidence of a physical and functional association between Cajal Bodies and PML Bodies. A direct interaction has been identified between coilin, the architect protein of the CB, and PIASy, an integral component of PML bodies, which could act as the molecular bridge linking these two defense centers. This connection suggests a flow of information or components between them. Moreover, the mouse Mx1 protein, the nuclear guardian against influenza, has been found associated with components of PML bodies, physically placing it at the heart of the nuclear antiviral response machinery.
The Cajal Body as an ISG Platform
This interconnected network reveals a new facet of the Cajal Body. It is not merely a passive target for viral hijacking, but also an active platform for the cellular counteroffensive. In an interferon-induced state of alert, the CB recruits effectors of innate immunity. A notable example is the exonuclease ISG20, another interferon-induced antiviral protein, which localizes predominantly in Cajal Bodies. This finding consolidates the view of the CB as an integrative node with a dual function. In times of peace, it is a factory for RNP biogenesis. In times of war, it becomes a militarized platform that assembles antiviral defense components. The nature of the CB as a liquid condensate is key to this functional plasticity. Its composition can change dynamically: in the absence of infection, it is enriched with RNA processing factors; following the interferon signal, it can recruit new components such as ISGs to shift its function. The cell does not need to build a new factory; it simply changes the personnel and tools in the one that already exists.
The Cajal Body as a Cellular Stress Sensor: A New Dimension in Immunity
The function of the Cajal Body transcends mere production of molecular components. Growing evidence suggests that both the CB and its architect protein, coilin, act as sensors of the cell’s state of health. Various types of cellular stress — such as UV radiation, osmotic stress, or DNA damage — provoke a rapid alteration in the structure of CBs and a redistribution of coilin and other components. This response is not a simple collapse, but an active reorganization believed to form part of a broader stress signaling network, in which the CB, in concert with the nucleolus, plays a central role.
Viral infection represents one of the most profound forms of cellular stress. From this perspective, the disruption of Cajal Bodies by viruses acquires a new meaning. It is not merely a matter of hijacking a factory, but of deactivating an alarm system. Coilin, in particular, has been shown to be involved in the regulation of immunity genes and in the activation of defense mechanisms, especially in response to viral infections. It has been observed that coilin is recruited to sites of DNA damage — a type of aggression that some viruses provoke — and that its presence modulates the repair response.
Therefore, it is plausible that the viral strategy of fragmenting or reorganizing CBs constitutes a counterintelligence tactic: by dismantling these detection and signaling centers, the virus not only appropriates their resources, but also blinds the cell to its own presence, sabotaging a coordinated immune response from within the nucleus itself. This view of the Cajal Body as a nexus of innate immunity — a guardian that is neutralized by the invader — adds a fascinating layer of complexity to the ceaseless battle waged within our cells.
From Observation to Therapy: New Antiviral Frontiers
The very characteristic that makes the Cajal Body a primary target for viruses — its central role in RNA metabolism — also makes it an exceptionally promising therapeutic target for the development of broad-spectrum antiviral drugs. If we can understand in detail how viruses hijack coilin and dismantle the CB, we can design molecular interventions to block this process and halt infection.
This idea is giving rise to a new paradigm in drug discovery: the development of “condensate-modifying drugs” (c-mods). Unlike traditional drugs, which typically inhibit the active site of a single enzyme following a “lock-and-key” model, these molecules would act by modulating the physical properties of the condensate as a whole. They could, for example, prevent its formation, force its dissolution, or alter its composition to make it inhospitable for the virus. The specific interaction between a viral protein and a scaffolding protein like coilin represents an ideal point of intervention.
The feasibility of this approach is not merely theoretical. Compelling proof of concept comes from the field of neurodegenerative diseases, particularly Spinal Muscular Atrophy (SMA). This devastating disease is caused by deficiency of the SMN protein, a fundamental component of the Cajal Body. The loss of Cajal Bodies is a distinctive cellular hallmark of SMA, and their number correlates inversely with disease severity, underscoring the critical importance of CB integrity for human health.
The extraordinary success of antisense oligonucleotide (ASO) therapies for SMA, such as Nusinersen, validates this approach. These drugs work by directly correcting the splicing process of the SMN2 gene, thereby increasing the production of functional SMN protein and partially restoring Cajal Body function. This is an example of a clinically successful therapy that works by manipulating the very same RNA processing machinery that is assembled and maintained in the CB.
There is a striking therapeutic convergence in the Cajal Body: a virus hijacks it to replicate, while a genetic disease arises from its dysfunction. A successful therapy for SMA works by restoring it. This demonstrates that the functional integrity of the CB is a pillar of cellular health. The clinical success in SMA provides a direct roadmap for the development of host-directed antivirals. It demonstrates that the molecular pathways converging on the CB are “pharmacologically accessible” and that protecting its integrity is a viable therapeutic strategy. The lessons learned from SMA can directly inform the design of a new generation of antivirals that seek to “armor” the CB against viral attack, potentially offering broad-spectrum protection with a new generation of antivirals.
The Perennial Relevance of a Sage
The story of the Cajal Body is an extraordinary journey that closes a perfect circle. The “accessory body” that Cajal’s artistic eye first captured in 1903 is today a dynamic center of cellular life and a nexus of innate immunity. The battle he waged against cholera in the streets of Valencia is now being fought, at the molecular level, against a myriad of viruses within that very structure he discovered.
Cajal’s genius was not limited to static anatomy; he was a theorist of function. His famous arrows, drawn by hand to indicate the direction of neural information flow, were the seed of the circuit theory upon which all neuroscience is based. In his work Textura del sistema nervioso, he went further, postulating that the pyramidal cells of the cerebral cortex were the “psychic cells,” the material substrate of the highest activities such as thought and memory. He intuited that the complexity of these cells was the basis of intelligence.
There is a beautiful conceptual symmetry between Cajal’s two great legacies. He conceptualized the brain as a network of discrete units (neurons) that process information. More than a century later, his other discovery, the CB, reveals itself as a node within a network of nuclear condensates that process genetic information. There is a kind of fractality in his vision: the principles of organization and information flow that he postulated for the macrocosm of the brain are reflected in the microcosm of the cell nucleus. The struggle for control of the Cajal Body is, in essence, a struggle for control of the cell’s information. The great unifying theme of Cajal’s work, in both neuroscience and cell biology, is the physical organization of biological information processing.
Cajal’s true legacy lies not solely in his discoveries, but in his scientific philosophy: a tireless curiosity, an unshakeable faith in meticulous observation, and the profound conviction that science must serve humanity. He believed that science was not a preserve reserved for geniuses, but the result of perseverance and “obstinate work” (“trabajo obstinado”). The researchers who today unravel the secrets of the Cajal Body are the direct heirs of this spirit. Their work demonstrates that, even in his most seemingly secondary observations, Cajal’s gaze was capable of pointing the way to the future. As he himself stated, the exploration of the biological universe is an endless enterprise. Research on the Cajal Body, a small world within the cell, is living proof of his vision.
“As long as our brain is a mystery, the universe, a reflection of the structure of the brain, will also be a mystery” (“Mientras nuestro cerebro sea un misterio, el universo, reflejo de la estructura del cerebro, sera tambien un misterio”).
Santiago Ramon y Cajal
Rock & Roll



Professor Angus Lamond and Cajal
The Lamond laboratory, whose team you can see on the website, is the creator of the video “A Christmas Cajal.” In this Christmas story, the key scientific moment is when they manage to record the movement of the Cajal body.
Bibliography
-
Ramon y Cajal, S. (1903). Un sencillo metodo de coloracion selectiva del reticulo protoplasmatico y sus efectos en los diversos organos nerviosos. Trabajos del Laboratorio de Investigaciones Biologicas de la Universidad de Madrid, 2, 129-221.
-
Ramon y Cajal, S. (1899-1904). Textura del sistema nervioso del hombre y de los vertebrados. Madrid: Moya.
-
Ramon y Cajal, S. (1917). Recuerdos de mi vida: Historia de mi labor cientifica. Madrid: Moya.
-
Ramon y Cajal, S. (1885). Investigaciones experimentales sobre el microbio virgula del colera y sus inoculaciones profilacticas. Zaragoza: Tipografia del Hospicio Provincial.
-
Gall, J. G. (2000). Cajal’s “accessory body”: the first hundred years. Annual Review of Cell and Developmental Biology, 16, 273-300.
-
Gall, J. G. (2003). The centennial of the Cajal body. Nature Reviews Molecular Cell Biology, 4(12), 975-980.
-
Banani, S. F., Lee, H. O., Hyman, A. A., & Rosen, M. K. (2017). Biomolecular condensates: a new cellular organizing principle. Nature Reviews Molecular Cell Biology, 18(5), 285-298.
-
Lettin, L., Erbay, B., & Blair, G. E. (2023). Viruses and Cajal Bodies: A Critical Cellular Target in Virus Infection?. Viruses, 15(12), 2311.
-
Haller, O., & Kochs, G. (2020). Human MxA protein: A key player in innate immunity to influenza A virus. Journal of Experimental Medicine, 217(1), e20190933.
-
Bhat, P., Aksenova, V., Gazzara, M. R., et al. (2023). Influenza virus mRNAs encode determinants for nuclear export via the cellular TREX-2 complex. Nature Communications, 14(1), 2304.
-
White, L., Erbay, B., & Blair, G. E. (2023). The Cajal body protein p80-coilin forms a complex with the adenovirus L4-22K protein and facilitates the nuclear export of adenovirus mRNA. mBio, 14(5), e01459-23.
-
Finkel, R. S., Mercuri, E., Darras, B. T., et al. (2017). Nusinersen versus Sham Control in Infantile-Onset Spinal Muscular Atrophy. New England Journal of Medicine, 377(18), 1723-1732.
-
Arranz-Herrero, J., Izpura-Luis, S., Presa, J., et al. (2025). Swine influenza-modified pulmonary microbiota. Frontiers in Cellular and Infection Microbiology, 15, 1634469.
-
DeFelipe, J. (2006). Brain plasticity and mental processes: Cajal again. Nature Reviews Neuroscience, 7(10), 811-817.
Un sencillo metodo de coloracion selectiva del reticulo protoplasmatico — Docs.Santiagoramonycajal
Trabajos del Laboratorio de Investigaciones Biologicas de la Universidad de Madrid = Travaux du Laboratoire de Recherches Biologiques de l’Universite de Madrid. SIMURG.
Trabajos del Laboratorio de Investigaciones Biologicas de la Universidad de Madrid = Travaux du Laboratoire de Recherches Biologiques de l’Universite de Madrid. Vol. 2. 990001153000204201_V02. — Docs.Santiagoramonycajal
Trabajos del Laboratorio de Investigaciones Biologicas de la Universidad de Madrid = Travaux du Laboratoire de Recherches Biologiques de l’Universite de Madrid. SIMURG.



